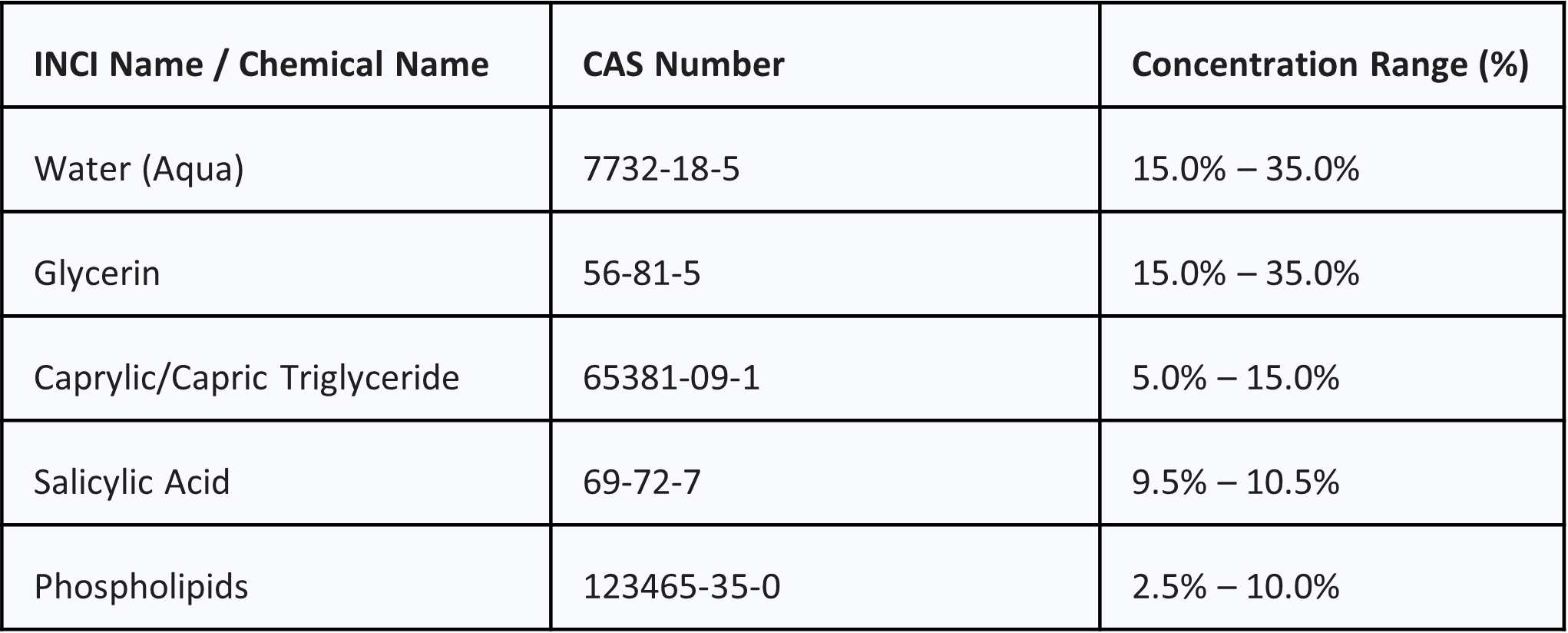
Product Overview
INCI Name
The INCI name is MyristylNicotinate, which is the standardized name in the cosmetic industry.
Product Type
It is an advanced bioactive ingredient specifically designed for skin and hair care products.
Key Feature
It can convert to NAD + in skin cells, improving penetration and reducing irritation compared to regular nicotinic acid.
Features & Applications
Applications
Mechanism of Action
The NAD + pathways and conversion process ofNanoActiveMN
NAD+ Pathways
NAD+ Importance
NAD + is essential for energy generation in mitochondria, lipid synthesis, DNA damage repair, and maintaining mitochondrial function and telomere length.
NAD+ Synthesis Pathways
There are three main pathways: the de novo pathway starting from tryptophan, the Preiss – Handler pathway using nicotinic acid, and the salvage pathway recycling NAD + metabolites.
NAD+ and Skin Health
NAD + is critical for cellular repair, energy metabolism, and combating age – related decline in skin. Its levels decrease by 50% by middle age.
NanoActiveMN Conversion
Efficacy Data
Barrier Repair:
Efficacy data on skin barrier repair
Anti – aging & Antioxidant
1. DPPH Radical Scavenging
The maximum clearance is 6.10% at a concentration of 0.3125%, showing its antioxidant ability.
2. ZO – 1 Protein Expression
At 0.04 mg/mL concentration, there is a 3.38 – fold relative expression of ZO – 1 protein.
3. Synergistic Effects with Retinoic Acid
It reduces irritation, enhances efficacy, and reduces water loss when used with retinoic acid.
Hair Loss Prevention
1. Clinical Study Design
A well – designed study was conducted to evaluate the efficacy ofNanoActiveMN on hair loss prevention.
2. Study Results
There was a significant improvement in hair abundance, density, quality, and thickness, with no adverse effects reported.
3. Mechanism of Action
It converts to NAD + in the scalp, enhancing cellular energy, improving barrier function, stimulating hair follicle cell renewal, and reducing oxidative stress.
Company Introduction
PuriPharmCo. Ltd.
1. Company Background
Specialized in bioactive ingredients, founded by industry experts and has global partnerships with research institutions.
2. Quality & Manufacturing
Strict quality control standards, state – of – the – art manufacturing facilities, and sustainable practices are in place.
3. R&D Capabilities
An expert technical team with PhD – level scientists and advanced laboratory facilities for cosmetic ingredient development.
Core Technologies
Nano Delivery:Advanced nanotechnology for enhanced bioavailability and targeted delivery of active ingredients.
Targeted Action:Allows for targeted action of ingredients at specific skin or hair sites.
Enhanced Penetration:Improves the penetration of ingredients into the skin for better efficacy.
Improved Stability: Ensures the stability of formulations during storage and use.
Formulation Stability
Stability information under different conditions
1. 24 – months Stability at Room Temperature
NanoActiveMN shows good stability in various formulations at room temperature for 24 months.
2. 3 – months Stability at 40℃
It has 3 – months stability at 40℃ in accelerated testing.
3. Minimal Degradation in Complex Formulations
There is minimal degradation in complex cosmetic matrices, ensuring long – term effectiveness.
Storage & Compatibility
Storage Conditions:
Store at 4 – 25℃ in a dry environment, protect from light, and keep the container tightly sealed.
Tested Formulations
It has been tested with various ingredients such as UV filters, emollients, humectants, natural oils, etc.
Ingredient Compatibility
Compatible with a wide range of cosmetic ingredients, including retinoids, antioxidants, emulsifiers, and thickeners.