1. PRODUCT OVERVIEW
NanoActive SA is an innovative, high-performance delivery system of Salicylic Acid designed specifically for advanced personal care and dermatological formulations. While Salicylic Acid (BHA) is renowned for its exceptional comedolytic and keratolytic properties, its application is often restricted by poor water solubility, crystallization risks, and potential skin irritation at efficacious doses.
PuriPharm utilizes state-of-the-art liposomal nano-encapsulation technology to entrap Salicylic Acid within a biomimetic phospholipid and triglyceride matrix. This proprietary delivery system not only renders the active ingredient highly dispersible in aqueous environments but also ensures a sustained-release profile. The result is a profoundly stabilized, highly bioavailable active that maximizes targeted efficacy while virtually eliminating the stinging and erythema traditionally associated with chemical exfoliants.
2. INCI / CHEMICAL DESCRIPTION
The following composition reflects the typical concentration ranges formulated to ensure optimal encapsulation and stability.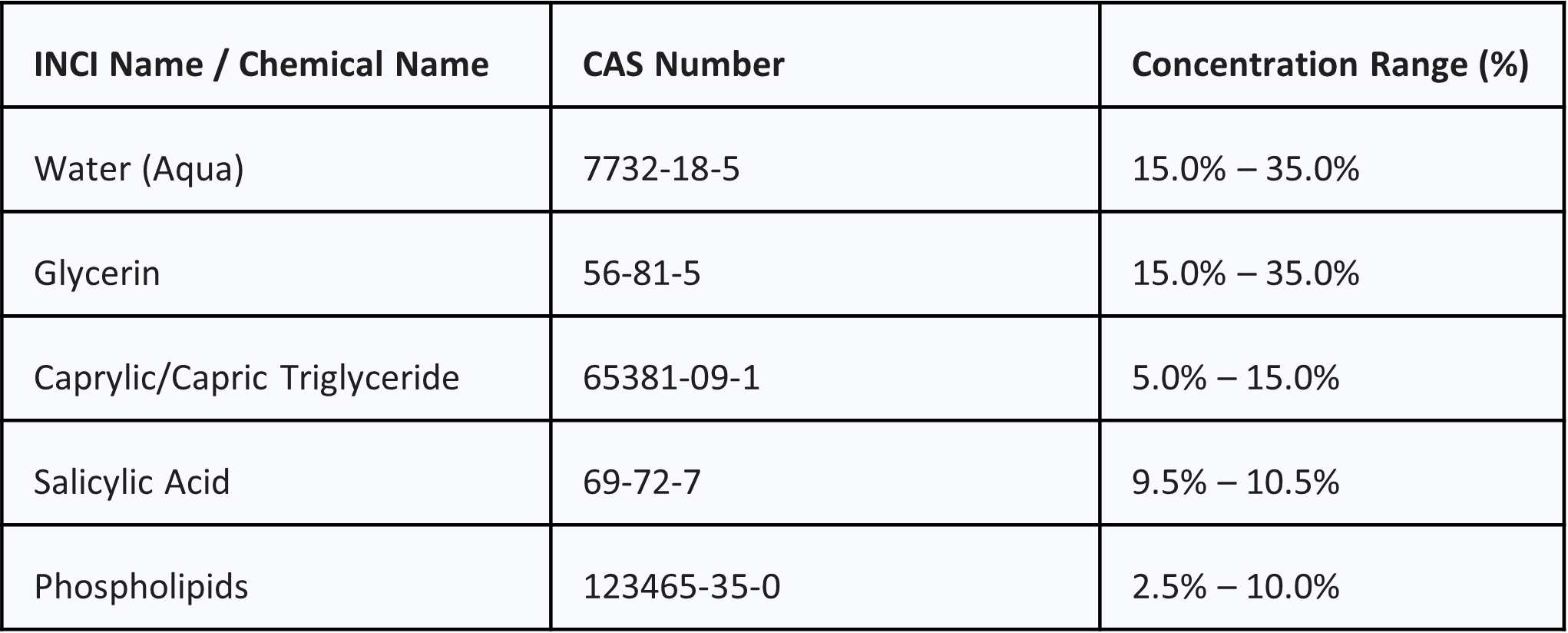
3. KEY FEATURES & ADVANTAGES
- Superior Water Dispersibility: Seamlessly incorporates into clear aqueous systems, toners, and gels without the need for high concentrations of harsh solvents (e.g., alcohol or excessive glycols).
- Biomimetic Lipid Matrix: Utilizing phospholipids and natural triglycerides, the carrier system exhibits excellent skin affinity, actively replenishing the stratum corneum while delivering the BHA.
- Controlled & Sustained Release: Modulates the delivery rate of Salicylic Acid, significantly mitigating acute epidermal shock and reducing potential erythema or stinging.
- Crystallization Prevention: The nano-matrix physically prevents Salicylic Acid molecules from aggregating and precipitating over time, ensuring long-term shelf stability of the finished cosmetic product.
4. MECHANISM & TECHNOLOGY HIGHLIGHTS
Targeted Follicular Delivery via Nano-vesicles: NanoActive SA functions by enclosing the lipophilic Salicylic Acid core within a phospholipid-based outer shell. When applied to the skin, these sub-micron liposomal carriers exhibit a high affinity for the lipid-rich environment of the pilosebaceous unit. Upon penetrating the pore, the matrix undergoes a gradual degradation process triggered by the skin’s natural physiological environment, releasing the active payload continuously over several hours. This approach ensures maximum keratolytic action exactly where acnegenesis occurs.
5. PHYSICAL & CHEMICAL PROPERTIES
Safety & Purity Specifications:
- Heavy Metals: Pb < 10 mg/kg | As < 2 mg/kg | Hg < 1 mg/kg | Cd < 5 mg/kg
- Microbiology: Total Aerobic Count < 100 CFU/g | Yeast & Mold < 10 CFU/g
- Diethylene Glycol: < 1000 mg/kg
6. SOLUBILITY & COMPATIBILITY
- Solubility: Readily dispersible in water, glycols, and standard aqueous media.
- Compatibility: Highly compatible with a wide range of cosmetic excipients, including non-ionic and anionic surfactants, and standard thickeners. Avoid combining with strong alkaline agents that may disrupt the phospholipid encapsulation matrix.
7. PRODUCT BENEFITS & COSMETIC FUNCTIONS
- Intensive Anti-Acne: Reduces sebum secretion, unplugs impacted pores, and inhibits Cutibacterium acnes proliferation.
- Gentle Exfoliation: Promotes the shedding of dead keratinocytes, resulting in a smoother, more radiant complexion.
- Pore Minimizing: Clears cellular debris from pores, visibly reducing their appearance.
- Scalp Care (Anti-Dandruff): Effective in removing scalp buildup and regulating scalp hyperkeratosis without stripping hair shafts.
8. RECOMMENDED APPLICATIONS
- Facial Skincare: Anti-acne serums, clarifying toners, exfoliating lotions, spot treatments, and deep-cleansing masks.
- Cleansers: Foaming gel cleansers and purifying washes.
- Hair & Scalp Care: Anti-dandruff shampoos and scalp purifying serums.
9. SUGGESTED USE LEVEL & REGULATORY STATUS
- Suggested Use Level: 0.1% – 20.0%(Applicable for both leave-on and rinse-off formulations).(Note: A 20% addition of NanoActive SA delivers 2.0% of pure Salicylic Acid).
- Regulatory Restriction: Due to the presence of Salicylic Acid, this product is not to be used in products intended for children under 3 years of age, with the exception of shampoos.
10. FORMULATION GUIDANCE
- Processing Phase: It is highly recommended to add NanoActive SA during the final cooling phase of the manufacturing process, ideally at temperatures below 45°C (113°F), to preserve the integrity of the liposomal structure.
- pH Range: Maintain the final formulation pH between 3.5 and 6.0for optimal efficacy and stability.
- Agitation: Stir gently but thoroughly. Avoid extreme high-shear homogenization post-addition.
11. STORAGE AND HANDLING
- Storage Conditions: Store in tightly closed original containers in a cool, well-ventilated, and dry place. Optimal storage temperature is between 15°C and 25°C.
- Light & Temperature: Protect from direct sunlight, prolonged heat exposure, and freezing.
- Handling: Handle in accordance with good industrial hygiene and safety practices. Refer to the Material Safety Data Sheet (MSDS) for detailed safety instructions.
12. SHELF LIFE
24 months from the date of manufacture when stored properly in unopened original packaging under recommended conditions.
13. PACKAGING INFORMATION
Standard pack sizes available:
- 1 kg / 5 kg plastic fluorinated jugs
- 25 kg tightly sealed HDPE drums(Customized packaging available upon request).
CONTACT INFORMATION
PuriPharm Co. Ltd.
Building 6, No. 1366 Hongfeng Road,
Huzhou, Zhejiang, China
Tel:+86-572-2745768 Email:service@puripharm.com
DISCLAIMER:
The information provided in this Product Data Sheet is based on our current knowledge and represents typical characteristics. It is offered in good faith but without warranty. It is the responsibility of the formulator and the user to ensure that the final cosmetic product is safe, stable, and complies with all applicable local regulations and patent laws. PuriPharm Co. Ltd. reserves the right to modify product specifications without prior notice.