Advanced Nano-Encapsulated Bio-Derived Sulfur for Multi-Targeted Skin Solutions
Company: PuriPharm Co. Ltd.
Website: www.PuriActives.com
Contact: Service@puripharm.com | Tel: 13167223168
1. PRODUCT OVERVIEW
Sulfur is a naturally occurring nonmetal with a long-standing dermatological history of effectively treating various skin disorders. However, traditional sulfur applications are severely limited by poor water solubility, large particle sizes (2–5 μm), low bioavailability, and formulation challenges.
NanoActive Sulfur, developed by PuriPharm Co. Ltd., is a cutting-edge active ingredient that utilizes a proprietary Nano-Delivery System (NDS). By stably encapsulating highly active bio-derived sulfur into sub-30 nm lipid-based nanocarriers, it completely resolves traditional solubility and aesthetic issues. The result is a highly bioavailable, transparent, and universally compatible active that regulates sebum, reduces inflammation, and offers potent antibacterial properties for acne and scalp care.
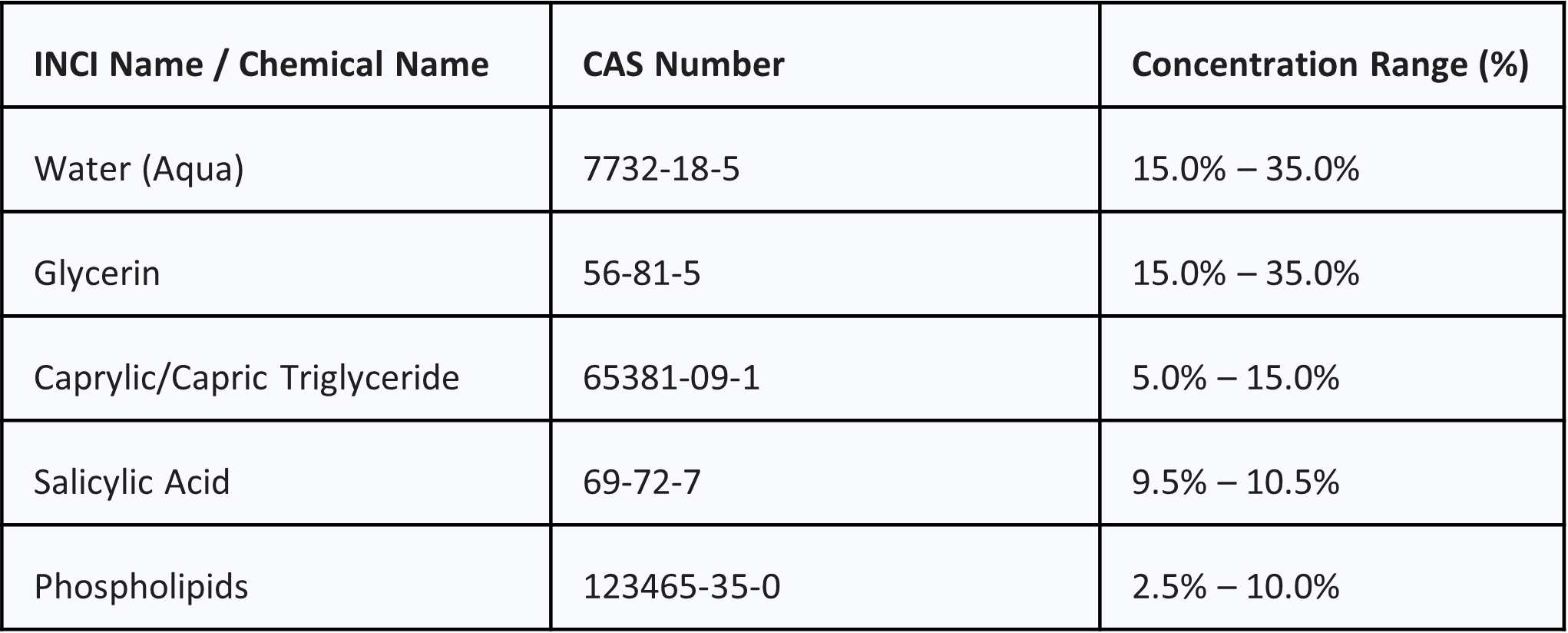
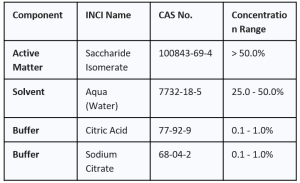
2. COMPOSITION & INCI INFORMATION
NanoActive Sulfur is formulated using a specialized biomimetic lipid matrix designed for optimal trans-epidermal delivery. It is strictly free from added preservatives and antioxidants.
Composition Range Table:
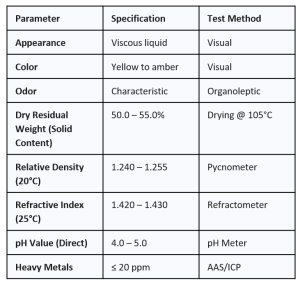
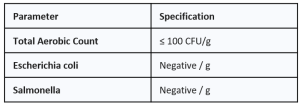
3. TECHNICAL SPECIFICATIONS
4. TECHNOLOGY HIGHLIGHTS: THE NDS ADVANTAGE
- Ultra-Fine Particle Size (< 30 nm): Significantly increases the specific surface area and interaction with keratinocytes, ensuring superior skin penetration and biological activity compared to traditional 2-5 μm sulfur.
- Exceptional Formulation Flexibility: The unique hydrophilic-lipophilic structural modification allows NanoActive Sulfur to be seamlessly incorporated into lotions, creams, clear gels, and water-based formulas without precipitation.
- Aesthetic Superiority: Unlike traditional sulfur, which is chalky and difficult to suspend, NanoActive Sulfur maintains optical clarity (forming a transparent solution at typical usage levels), leaving no residue and enabling elegant cosmetic formulations.
- High Stability & Bioavailability: The robust phospholipid-based nanocarrier prevents aggregation, protects the active payload, and ensures targeted delivery to the sebaceous glands and follicles.
5. KEY COSMETIC BENEFITS & MECHANISM OF ACTION
A. Comprehensive Sebum Regulation & Keratolysis
Excessive sebum accumulation leads to enlarged pores, follicular blockage, and inflammation. NanoActive Sulfur effectively removes surface lipids, softens the epidermis, and dissolves keratin plugs.
●Mechanism:At low concentrations, sulfur exerts a keratoplastic effect, promoting normal keratinization. At higher concentrations, it becomes keratolytic, reacting with cysteine to form cystine and hydrogen sulfide (), which safely disrupts excess keratin and clears clogged pores.
B. Targeted Antibacterial & Antifungal Action
NanoActive Sulfur acts as a potent purifying agent and helps rebalance the skin microbiome.
●Mechanism: Skin bacteria and keratinocytes convert sulfur into pentathionic acid (), a compound highly toxic to fungi and pathogenic bacteria. The keratolytic action further helps detach microorganisms from the stratum corneum.
●Microbiological Efficacy (In Vitro):
○P. aeruginosa: MIC = 1.95 μg/mL | MBC = 1000 μg/mL
○S. aureus: MIC = 125 μg/mL
○E. coli: MIC = 500 μg/mL
C. Powerful Anti-Inflammatory & Soothing Properties
Severe oxidative stress depletes intracellular Glutathione (GSH), triggering inflammatory cascades.
●Mechanism:NanoActive Sulfur supports cellular antioxidant defenses by promoting GSH synthesis.
●Efficacy:Exerts a strong, dose-dependent reduction in the expression of key pro-inflammatory cytokines. Clinical evaluations (ELISA) show that concentrations between 0.1% and 0.4% significantly downregulate the protein expression ofIL-1β,IL-6, and TNF-α.
D. Clinically Proven Acne Treatment
Inin-vivoassessments, formulations containing NanoActive Sulfur demonstrated visible improvement in acne-prone skin profiles. A 60-day application of 0.2% active showed a significant reduction in inflammatory lesions and post-acne erythema, making it an ideal candidate for long-term acne management.
6. RECOMMENDED APPLICATIONS
NanoActive Sulfur is highly versatile and is recommended for the following cosmetic product categories:
- Acne & Blemish Control: Spot treatments, anti-acne serums, and purifying toners.
- Sebum Control & Pore Refining: Mattifying lotions, oil-control powders, and deep-cleansing masks.
- Brightening & Spot-Fading: Post-inflammatory hyperpigmentation (PIH) correctors.
- Scalp Care: Anti-dandruff shampoos and scalp-soothing serums (seborrheic dermatitis support).
- Soothing & Repair: Products targeting sensitized, inflamed, or compromised skin barriers.
7. FORMULATION GUIDANCE
- Suggested Use Level: 0.1% – 5.0% (Depending on target efficacy: low doses for daily maintenance/anti-inflammation; higher doses for targeted spot treatments).
- Solubility: Amphiphilic nature allows it to be easily added to either the aqueous phase or the final emulsion at room temperature or gently heated conditions.
- Compatibility: Compatible with a wide range of cosmetic ingredients, including traditional water-based and oil-based systems. Does not cause phase separation.
8. STORAGE, HANDLING & PACKAGING
- Storage: Store in the original, unopened container at 4°C to 25°C. Protect from direct sunlight and maintain in a dry environment.
- Shelf Life: 12 months under specified storage conditions.
- Packaging: Supplied in standard industrial plastic containers.
Disclaimer: The information contained in this document is provided in good faith and is based on our current knowledge and internal testing. Formulators are responsible for ensuring the final cosmetic product’s stability, safety, and regulatory compliance in their respective target markets.
PuriPharm Co. Ltd. | Advanced Active Ingredients